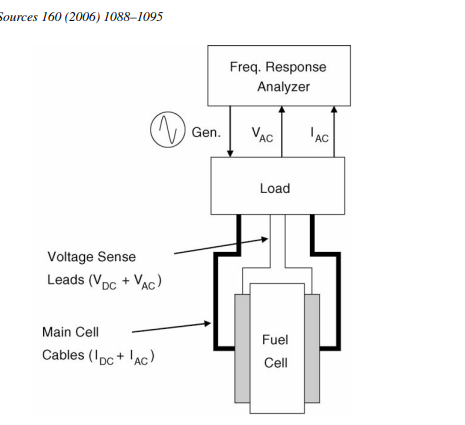
Electrochemical Impedance Spectroscopy
The most common method of EIS is the semicircular Nyquist plot shown in the figure below (the terminology of Nyquist plot will be explained later).
Why does it appear so often? Much of the usual electrochemistry deals with heterogeneous systems, that is, charge transfer phenomena at the electrode-solution interface. At the heterogeneous interface, a double layer with pairs of positive and negative charges is formed. That is, electrical capacity, which requires activation for charge transfer, which corresponds to an electrical resistance. These two generally occur simultaneously in parallel, and not in a sequential process, one after the other. There are so many states and phenomena that are replaced by parallel circuits (simultaneous processes) of resistance and capacitance.
In the figure on the right, an equivalent circuit is inserted to help understanding (the equivalent circuit is an auxiliary measures for intuitively interpreting EIS by replacing phenomena and processes with electronic circuit elements). It is an equivalent circuit consisting of a parallel circuit of capacitance C and resistance R and resistance Rs in series. If we assume that C is a capacitor derived from the electrode interface double layer, the parallel resistances are considered to be charge transfer resistances derived from the electrode active material. The faster the charge transfer rate, the smaller the resistance will be. The charge transfer rate depends on various factors such as the type and concentration of the active material, the potential of the electrode, and the way it is supplied. This corresponds to the uncompensated solution resistance that the potentiostat cannot compensate, as the solution resistance between the working and reference electrodes. In other words, it is a common equivalent circuit of an electrochemical system. It has been called as Randles circuit.